- Blog
- Realistico full free
- Review jixipix watercolor studio
- How to use depriester chart
- Spotflux 2-10-7
- Shanky bot profiles
- Mx vs atv untamed cheats ps2
- Opera women
- Fortress of fear game boy
- Henry iv part 1 folger
- Cxbx reloaded halo
- Famous photographers portraiture
- Marlow briggs and the mask of death
- -parentkey error prodiscover basic

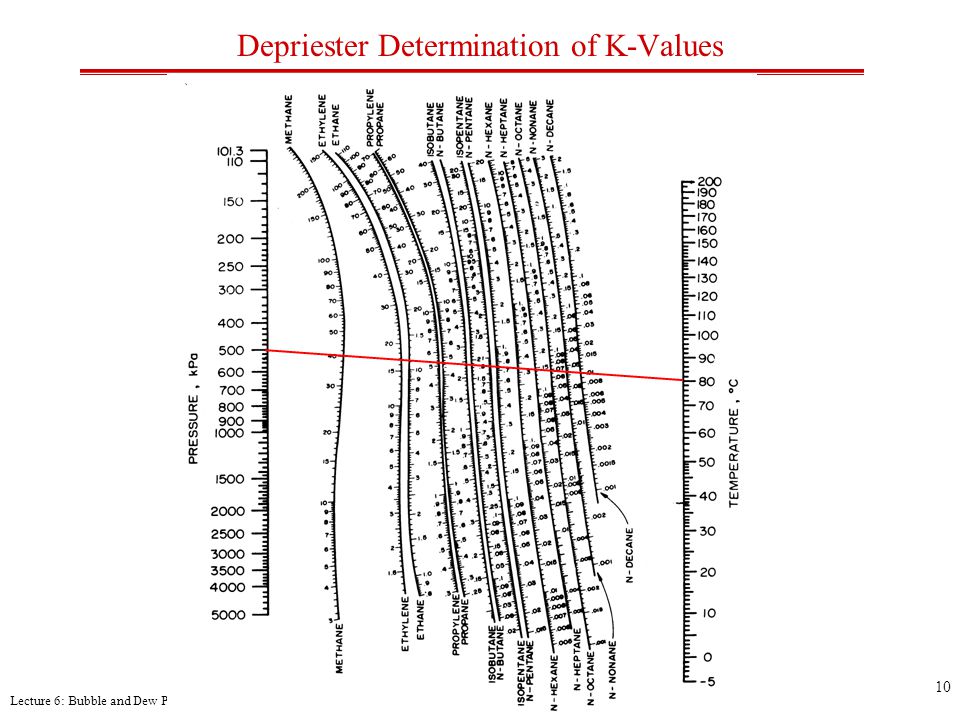
SI versions of these charts have been developed by Dadyburjor. For K as a function of T and P only, the DePriester charts provide good starting values for the iteration. One cannot calculate K values until phase compositions are known, and those cannot be known until the K values are available to calculate them. The Kellogg charts, and hence the DePriester charts, are based primarily on the Benedict-Webb-Rubin equation of state, which can represent both the liquid and the vapor phases and can predict K values quite accurately when the equation constants are available for the components in question.Ī trial-and-error procedure is required with any K-value correlation that takes into account the effect of composition. These charts are a simplification of the Kellogg charts and include additional experimental data. The easiest to use are the DePriester charts, which cover 12 hydrocarbons (methane, ethylene, ethane, propylene, propane, isobutane, isobutylene, n-butane, isopentane, n- pentane, n-hexane, and n-heptane). For example, several major graphical K-value correlations are available for light-hydrocarbon systems. However, for mixtures of compounds of similar molecular structure and size, the K value depends mainly on temperature and pressure. 4, the K value of a species is a complex function of temperature, pressure, and equilibrium vapor- and liquid-phase compositions.
- Blog
- Realistico full free
- Review jixipix watercolor studio
- How to use depriester chart
- Spotflux 2-10-7
- Shanky bot profiles
- Mx vs atv untamed cheats ps2
- Opera women
- Fortress of fear game boy
- Henry iv part 1 folger
- Cxbx reloaded halo
- Famous photographers portraiture
- Marlow briggs and the mask of death
- -parentkey error prodiscover basic
